
By Jeff Kessen
Lead-acid batteries have long been the default backup power choice for the uninterruptible power supplies (UPS) that ensure the availability of data centers, communications equipment, and industrial processes. While they provide the power and reliability required for these applications, when supported with appropriate monitoring and maintenance practices, they have traditionally been regarded as the weak link in the critical power chain. They tend to be high maintenance, heavy and require frequent replacement.
Now, lithium-ion batteries have emerged as a viable alternative and a growing number of users are evaluating this technology for UPS applications in mission-critical environments.
One of the key attractions for moving to lithium-ion batteries from lead-acid batteries is the extended battery life. Yet, with limited operating data in UPS applications available today, it’s natural for potential users to question how long lithium-ion batteries will actually last.
To address this question, it’s necessary to first understand how lithium-ion batteries degrade under normal circumstances. They have two independent modes of degradation: calendar life and cycle life.
Calendar life describes how the capacity will decline and how the resistance will increase over time. For calendar life, the operating temperature of the battery is the most important factor in determining how long it will last. Heat can accelerate degradation and cooler temperatures minimize degradation.
As for cycle life, the term itself is easily understood but projecting cycle life of a lithium-ion battery is not as simple as it would seem. This is because different characteristics of the cycle determine how damaging that cycle is to the battery.
In data center applications, calendar life is typically the primary driver because battery cycling is infrequent. The exception to this would be when UPS batteries are being used to support site energy management or providing grid services to the local electricity distribution network. Those applications are outside the scope of this paper. In conventional use cases, the batteries are idle for most of their operating life.
To assess calendar life, cell manufacturers typically store batteries at different temperatures for long periods of time and periodically check their remaining capacity. Charting this data provides a relationship between time, temperature and remaining capacity. After enough data has been collected, it’s possible to fit the data to a generally accepted equation for calendar life which has been proven over decades of lithium-ion battery field experience. An example of a lithium-ion battery calendar life chart is shown in Figure 1.
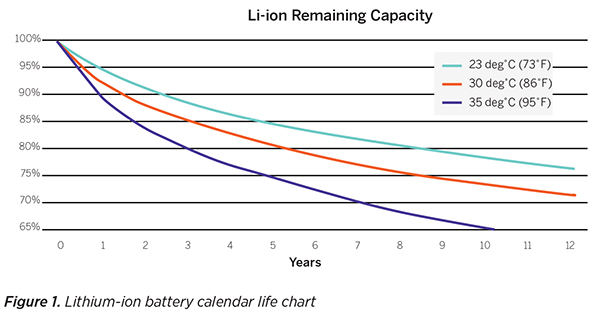
Figure 1 shows that 80 percent of initial capacity remains after about ten years at temperatures of 23°C (73°F) for the particular lithium-ion battery tested. Different lithium-ion batteries will have different rates of degradation. What is important is that 80 percent capacity is not a significant milestone in the life of a lithium-ion battery because the rate of annual degradation is slowing down around 80 percent and will continue to degrade predictably. The next section will cover how the remaining capacity and battery resistance influence the runtime in a specific application.
PROJECTING FUTURE RUNTIMES FOR LITHIUM-ION BATTERIES
One of the most common questions potential users of lithium-ion batteries have concerns the impact of battery age on projected run times. This concern is reflected in a common question: Is a lithium battery susceptible to surprise failures late in life similar to lead-acid batteries?
To address this question, it’s important to consider the previous discussion on lithium-ion battery degradation as well as the design of the electrodes in each cell of the battery system. A lithium-ion battery cell can be tailored to different performance objectives by the manufacturer and one key trade-off is between how fast you can charge or discharge the battery and how much energy it holds. The extreme ends of these trade-offs are summarized in Figure 2.
| Performance Metric | Cell optimized for energy density | Cell optimized for power |
|---|---|---|
| Energy Density | Today’s lithium-ion technology can produce cell energy densities as high as 600 Wh/liter, which enables a very small footprint, but these batteries can’t discharge their energy very quickly. | Cells optimized for power performance make compromises on energy density. In extreme cases, the energy density of these cells can be just half of the high energy alternatives. |
| Rate Capabiltiy | A cell optimized for energy density may take an hour or more to fully discharge at its fastest sustainable rate. | Cells optimized for power can achieve full discharge in a few minutes without damaging the battery. |
| Electrical resistance | The internal resistance of these batteries is considerably lower than lead-acid batteries but high compared to a lithium-ion power battery. If they are charged or discharged at their maximum rate, more heat will be generated. | The electrode design parameters that lead to high power capability also yield very low cell resistance so these batteries generate less heat, even during fast cycling. |
| Relative cost | These Cells are less expensive to manufacture and are generally produced in higher volumes. Market reports touting lower battery prices in terms of $ /kWh are almost always referring to batteries of this type. | The electrodes in high power cells are more difficult to manufacture and have additional cost drivers in the design. High-power batteries are typically much more expensive for the same amount of capacity (measured in kWh.) |
| Figure 2. Trade-oofs involved in optimizing lithium-ion cells for power and density | ||
For a UPS application, the key design objective for the battery manufacturer is to provide the required runtime at the lowest possible cost. As a result, most UPS solutions in the market today tend to use cells that are more optimized for energy density and cost and don’t usually employ cells that are inherently more capable of fast discharges. This also means that the chosen lithium-ion cells don’t have the lowest possible internal resistance and tend to experience considerable temperature increases when they are discharged in less than 10 minutes.
When analyzing runtimes for a lithiumion battery in a UPS application, it’s important to understand the characteristics of the cells being used because the end of discharge could be caused by the battery reaching minimum voltage, the battery reaching maximum temperature or the battery running out of energy due to its capacity limitations. In many cases, the condition that causes the discharge to end is also different at different loads.
Projecting runtime ten years in the future adds yet another level of difficulty. Contrary to popular belief, remaining capacity in the battery as it ages may not be what limits your runtime, particularly at high loads. In addition to losing capacity, lithium-ion batteries also experience increases in their internal resistance over their life and this leads to larger voltage drops and more self-heating than when they were new. As a result, the battery manufacturer should know not only how capacity declines but also how resistance increases according to the battery’s operating history and number of years in service.
Although runtime analysis for lithium-ion batteries can be complex to get right, users generally don’t face the risk that the battery might suddenly give them half the runtime they were expecting or were promised by the specifications. According to IEEE recommended practice on VRLA batteries (1188), lead-acid batteries need to be replaced when they reach 80 percent of their original capacity. That’s because the remaining capacity falls off very quickly after that point. That failure mode doesn’t happen with lithium-ion technology which tends to fade gradually and predictably.
LITHIUM-ION BATTERY SAFETY
Lithium-ion batteries have a few known conditions that can lead them to ignite or release gases if the internal pressure gets too high. Knowing these risk conditions and controlling them is the purpose of the battery management system (BMS).
For any product with safety relevance, good system integration practice starts with understanding the probability and severity of each potential failure mode. In the case of lithium-ion batteries, the riskiest abuse conditions are generally overcharge, overheating and short-circuiting of the battery cells. Each of these conditions can cause the electrolyte in a lithium-ion battery to decompose into gases or, in extreme cases, to ignite.
While some battery manufacturers make claims about the safety of their batteries based on the chemistry used in the cathode or anode, lithium-ion battery failures almost always start in the electrolyte and virtually all cells that are commercially available today use a flammable electrolyte. This is not to say that the chemistry of the cathode and anode do not influence battery safety. Those material choices can drive significant differences in the intensity of a failure, but it would be wrong to suggest that any lithium-ion cell is immune to the risks of abusive operating conditions.
The primary purpose of the BMS is to implement controls that keep the battery within its safe operating range. This involves continuous measurement of cell voltages, system temperatures and battery current in addition to other parameters. When the BMS senses that the battery is approaching one of its operating limits, a warning is communicated to the connected power device such as a UPS. If the UPS doesn’t react appropriately to keep the battery within its permissible operating boundaries, the BMS can disconnect the battery from the load or charger to maintain safety. The functionality and effectiveness of the BMS itself are also verified by the testing necessary to achieve UL or CE certification of the battery system. Stationary battery applications also benefit from the safety developments already driven by the auto industry.
Some operating thresholds in the BMS also consider more than just safety. In UPS applications, the thermal limit of the battery is a good example. Generally speaking, the BMS in data center applications has a temperature limit which is well below the level necessary to ensure safety because life considerations are also involved in setting that threshold. Therefore, a case of the BMS disconnecting a battery does not necessarily mean that safety was in question.
While a well-engineered BMS provides a high degree of confidence regarding lithium-ion battery safety, the one significant risk that it can’t control is the battery being in the presence of fire. It’s impossible to guarantee that a lithium-ion battery cabinet will never be in a building that catches fire for reasons that have nothing to do with the battery. This reality has recently driven the considerable expansion of fire codes related to lithium-ion batteries.
LITHIUM-ION BATTERY MONITORING
The BMS in a lithium-ion battery system is continuously monitoring numerous operating parameters to ensure safety. Since all this operating data is already stored in BMS memory, it is relatively simple for the BMS manufacturer to make the data available to external systems via a Modbus IP connection or some similar data acquisition protocol. This is a key advantage of lithium-ion batteries over lead-acid systems where any monitoring system needs to have its sensors installed.
MAKING THE MOVE TO LITHIUM-ION BATTERIES
Lithium-ion batteries have reached a stage of maturity where they can be considered as a viable replacement for lead-acid batteries in UPS applications. Despite the lack of operating data available, calendar life testing indicates that lithium-ion batteries can deliver a significantly longer life than lead-acid batteries without an increased risk of failure when capacities fall below 80 percent. In addition to ensuring safety, lithiumion battery management systems can help maximize battery life and minimize downtime by enabling continuous monitoring that supports condition-based maintenance. An experienced integrator can help you evaluate the total cost of ownership for lithium-ion batteries compared to lead-acid batteries for a particular application.
Jeff Kessen is Vice President of Energy Storage for Vertiv. He can be reached at [email protected].
Read Other Articles
(You Must Be Logged In to Continue…)